Researchers from the University of Tsukuba have categorized barthelonids, a group of microscopic anaerobic flagellates, as an early branching Metamonada lineage based on their genetic identity and mitochondrial evolutionary history
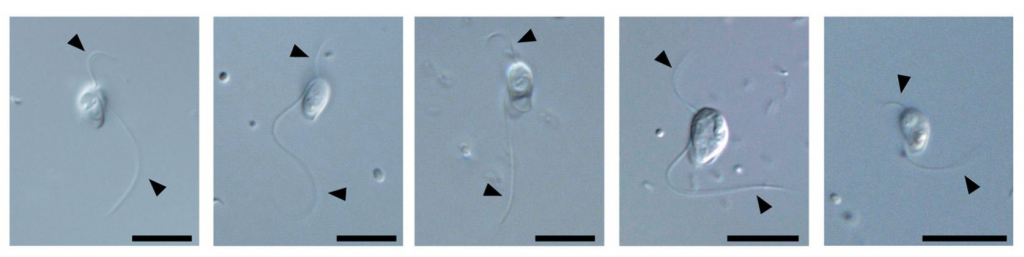
Tsukuba, Japan – New species of microbial life are continually being identified, but localizing them on a phylogenetic tree is a challenge. Now, researchers at the University of Tsukuba have pinpointed barthelonids, a genus of free-living heterotrophic biflagellates typified by Barthelona vulgaris, and clarified their ancestry as well as evolution of their ATP-generation mechanisms.
A phylogenetic tree portrays species by lineage. The trunk represents a common ancestor and the branches all its evolutionary descendants; together, a monophyletic group or clade. The eukaryotic Tree of Life represents the phylogeny of all organisms with nucleated cells, ranging from unicellular protists to blue whales. Where would the barthelonids fit?
The researchers established five strains of Barthelona species from different parts of the world. Analysis of the transcriptome of one strain (PAP020), its RNA “signature,” localized it on the phylogenetic tree to the base of the Fornicata clade. This indicated that the last common ancestor of the barthelonids evolutionarily diverged very early in the evolution of Metamonada.
Senior author Professor Yuji Inagaki explains: “We analyzed small subunit ribosomal DNA as well as phylogenomic data to confirm the commonality of all Barthelona strains. In order to deduce their phylogenetic position, we matched transcriptome data from PAP020 against a eukaryote-wide dataset containing 148 genes.”
The transcriptome data of PAP020 also indicated the evolutionarily adapted metabolic pathways of ATP generation. The research team suspected that barthelonids, being anaerobic, possessed mitochondrion-related organelles (MROs) instead of full-fledged mitochondria, a suspicion upheld by electron microscopy. Comparison with MROs in fornicates predicted that PAP020 could not generate ATP in the MRO, as no mitochondrial/MRO enzymes involved in substrate-level phosphorylation were detected. However, PAP020 possesses a cytosolic ATP synthase, acetyl-CoA synthetase (ACS), suggesting that PAP020 generated ATP in the cytosol.
“We have furthered current hypotheses around the evolutionary history of ATP-generating mechanisms in the Fornicata clade in light of data from Barthelona strain PAP020,” says Professor Inagaki. “Interestingly, the sequence ACS2 was formerly believed to be acquired at the base of the Fornicata clade, but we propose that this event occurred earlier with the common ancestor of fornicates and barthelonids. Indeed, it may have occurred further back with the last common metamonad ancestor. Loss of substrate-level phosphorylation from the MRO in the clade containing barthelonids with other fornicates could well be two discrete events.”
Original Paper
The article, “Barthelonids represent a deep-branching metamonad clade with mitochondrion related organelles predicted to generate no ATP” was published in Proceedings of the Royal Society B at DOI: http://doi.org/10.1098/rspb.2020.1538
